Predicting Carbon Monoxide Gas Buildup
Apps.CarbonMonoxide History
Show minor edits - Show changes to markup
Part 1: Derivation of Material Balance
Part 1: Derivation of CO Material Balance
Part 2: Numerical Solution of CO Material Balance
(:html:) <iframe width="560" height="315" src="https://www.youtube.com/embed/3yj_8dWd1VY?rel=0" frameborder="0" allowfullscreen></iframe>(:htmlend:)
Carbon monoxide poisoning is potentially fatal. Carbon monoxide sensors can provide alarms for buildup in areas where there is a likely source of emission such as from a flame source or internal combustion engine. In this situation, a 200 square foot room with 9 foot high ceilings has a 0.01 L/sec leak of CO gas. The air in the room is assumed to be well mixed and at constant temperature (300 K). Additionally, the volume and number of moles of gas in the room is constant. There are no open windows or doors but the gas that is added to the room displaces some of the well-mixed air out of the room.
Carbon monoxide poisoning is potentially fatal. Carbon monoxide sensors can provide alarms for buildup in areas where there is a likely source of emission such as from a flame source or internal combustion engine. In this situation, a 200 square foot room with 9 foot high ceilings has a 0.1 mL/sec leak of CO gas. The air in the room is assumed to be well mixed and at constant temperature (300 K). Additionally, the volume and number of moles of gas in the room is constant. There are no open windows or doors but the gas that is added to the room displaces some of the well-mixed air out of the room.
Below is a video tutorial that demonstrates how to predict the time evolving concentration of CO in parts per million in the room. A material balance on the CO gas entering, accumulating, and leaving the room volume is performed. This material balance results in a differential equation. An exact solution is obtained through a method of separation and integration, Laplace transforms, and with a numerical solver. Numerical solution is performed with MATLAB and Python. Example files are available for download below.
Below is a video tutorial that demonstrates how to predict the time evolving concentration of CO in parts per million in the room. A material balance on the CO gas entering, accumulating, and leaving the room volume is performed. This material balance results in a differential equation. A solution is obtained through a method of separation and integration, Laplace transforms, and with a numerical solver. Numerical solution is performed with MATLAB and Python. Example files are available for download below.
Part 1: Derivation of Material Balance
(:html:) <iframe width="560" height="315" src="https://www.youtube.com/embed/M52S1XiIIEo?rel=0" frameborder="0" allowfullscreen></iframe> (:htmlend:)
Carbon monoxide poisoning is potentially fatal. Carbon monoxide sensors can provide alarms for buildup in areas where there is a likely source of emission such as from a flame source or internal combustion engine. In this situation, a 200 square foot room with 9 foot high ceilings has a 0.01 L/sec leak of CO gas. The air in the home is assumed to be well mixed and at constant temperature (300 K). Additionally, the volume and number of moles of gas in the room is constant. There are no open windows or doors but the gas that is added to the room displaces some of the well-mixed air out of the room.
Carbon monoxide poisoning is potentially fatal. Carbon monoxide sensors can provide alarms for buildup in areas where there is a likely source of emission such as from a flame source or internal combustion engine. In this situation, a 200 square foot room with 9 foot high ceilings has a 0.01 L/sec leak of CO gas. The air in the room is assumed to be well mixed and at constant temperature (300 K). Additionally, the volume and number of moles of gas in the room is constant. There are no open windows or doors but the gas that is added to the room displaces some of the well-mixed air out of the room.
Below is a video tutorial that demonstrates how to predict the time evolving concentration of CO in parts per million in the room. A material balance on the CO gas entering, accumulating, and leaving the home is performed. This material balance results in a differential equation. An exact solution is obtained through a method of separation and integration, Laplace transforms, and with a numerical solver. Numerical solution is performed with MATLAB and Python. Example files are available for download below.
Below is a video tutorial that demonstrates how to predict the time evolving concentration of CO in parts per million in the room. A material balance on the CO gas entering, accumulating, and leaving the room volume is performed. This material balance results in a differential equation. An exact solution is obtained through a method of separation and integration, Laplace transforms, and with a numerical solver. Numerical solution is performed with MATLAB and Python. Example files are available for download below.
Carbon monoxide poisoning is potentially fatal. Carbon monoxide sensors can provide alarms for buildup in areas where there is a likely source of emission such as from a flame source or internal combustion engine. In this situation, a 2000 square foot home with 9 foot high ceilings has a 0.1 L/sec injection of CO gas. The air in the home is assumed to be well mixed and at constant temperature. Additionally, the volume and number of moles of gas in the home is constant. There are no open windows or doors but the gas that is added to the home displaces some of the well-mixed air out of the home.
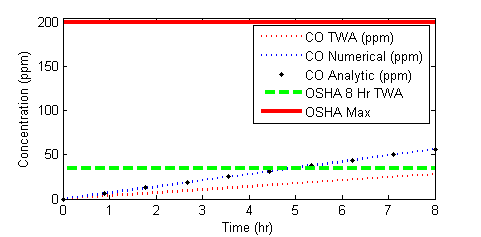
OSHA standards are a time weighted average of 35 parts per million (PPM) over an 8 hour work day and a maximum exposure level of 200 PPM. It is desired to calculate the concentration of CO in the home over an 8 hour period.
Below is a video tutorial that demonstrates how to predict the time evolving concentration of CO in parts per million in the home. A material balance on the CO gas entering, accumulating, and leaving the home is performed. This material balance results in a differential equation. An exact solution is obtained through a method of separation and integration, Laplace transforms, and with a numerical solver. Numerical solution is performed with MATLAB and Python. Example files are available for download below.
Carbon monoxide poisoning is potentially fatal. Carbon monoxide sensors can provide alarms for buildup in areas where there is a likely source of emission such as from a flame source or internal combustion engine. In this situation, a 200 square foot room with 9 foot high ceilings has a 0.01 L/sec leak of CO gas. The air in the home is assumed to be well mixed and at constant temperature (300 K). Additionally, the volume and number of moles of gas in the room is constant. There are no open windows or doors but the gas that is added to the room displaces some of the well-mixed air out of the room.
OSHA standards are a time weighted average of 35 parts per million (PPM) over an 8 hour work day and a maximum exposure level of 200 PPM. It is desired to calculate the concentration of CO in the room over an 8 hour period.
Below is a video tutorial that demonstrates how to predict the time evolving concentration of CO in parts per million in the room. A material balance on the CO gas entering, accumulating, and leaving the home is performed. This material balance results in a differential equation. An exact solution is obtained through a method of separation and integration, Laplace transforms, and with a numerical solver. Numerical solution is performed with MATLAB and Python. Example files are available for download below.
(:title Predicting Carbon Monoxide Gas Buildup:) (:keywords nonlinear, model, APMonitor, carbon monoxide, OSHA, nonlinear dynamics, mixing, pollutant:) (:description Determine carbon monoxide buildup in a room through the solution of a dynamic material balance:)
Carbon Monoxide Buildup
Carbon monoxide poisoning is potentially fatal. Carbon monoxide sensors can provide alarms for buildup in areas where there is a likely source of emission such as from a flame source or internal combustion engine. In this situation, a 2000 square foot home with 9 foot high ceilings has a 0.1 L/sec injection of CO gas. The air in the home is assumed to be well mixed and at constant temperature. Additionally, the volume and number of moles of gas in the home is constant. There are no open windows or doors but the gas that is added to the home displaces some of the well-mixed air out of the home.
OSHA standards are a time weighted average of 35 parts per million (PPM) over an 8 hour work day and a maximum exposure level of 200 PPM. It is desired to calculate the concentration of CO in the home over an 8 hour period.
Below is a video tutorial that demonstrates how to predict the time evolving concentration of CO in parts per million in the home. A material balance on the CO gas entering, accumulating, and leaving the home is performed. This material balance results in a differential equation. An exact solution is obtained through a method of separation and integration, Laplace transforms, and with a numerical solver. Numerical solution is performed with MATLAB and Python. Example files are available for download below.